Collaborative Projects
Collaboration lies at the heart of Swiss Biobanking. In a landscape where shared resources and collective effort drive scientific progress, we work closely with partners across Switzerland and Europe—through our BBMRI-ERIC membership—to serve the biobanking community. Collaborative projects with diverse partners—from research infrastructures and biobanks to academic and industry actors—strengthen our visibility and credibility and reflect our commitment to openness, partnership, and impact.

Swiss Projects
CPCR service finder
The CPCR Service Finder, a new collaborative, user-friendly tool, has been developed by the national Coordination Platform Clinical Research with the technical expertise of Swiss Biobanking. It allows to browse among services from the main national research infrastructures in one click, and to identify whom to approach for support.
As a first tangible outcome of the CPCR—whose mission is to foster synergies among organizations conducting and supporting clinical research in Switzerland—the CPCR Service Finder supports academic researchers, study coordinators, data managers, and biobank professionals in identifying which national infrastructures can provide relevant expertise, data, or resources at different stages of their projects.
The tool is the result of a collaboration within the national Coordination Platform Clinical Research (CPCR). It was developed in a joint effort of Swiss Biobanking (technical lead), Swiss Cancer Institute, Swiss Clinical Trial Organisation (SCTO) and Swiss Personalized Health Network (SPHN), with the support and coordination of the SAMS. swissethics joined as first partner to include its resources. The tool is designed to welcome other key national service providers progressively.
Who is it for?
The CPCR Service Finder is designed for anyone involved in health and clinical research in Switzerland, from academic researchers and study coordinators to data managers and biobank professionals. It supports users in identifying which national infrastructures can provide the expertise, data, or resources they need at different stages of their projects.
UMZH biobank
The University Medicine Zurich (UMZH) biobank initiative aims to strengthen coordination and visibility of biological samples across the Zurich academic medical ecosystem. In alignment with the vision of the LOOP Zurich, which promotes data-driven biomedical research through access to large clinical datasets and biobanks, the initiative seeks to make sample information from participating institutions discoverable and interoperable.
This effort is closely connected to the LOOP Biomedical Informatics Platform, a central infrastructure integrating clinical data from Zurich university hospitals and enabling researchers to identify and request available datasets in a legally, ethically, and technically compliant framework. Within this context, Swiss Biobanking Platform supports the development of the UMZH biobank ecosystem through three main pillars: (i) strengthening quality and governance through its national biobank labelling programme, (ii) improving the visibility of biological samples and biobanks through integration into its national Catalog, and (iii) enabling interoperability and data standardization through the implementation of a Biobank Information Management System (BIMS) and the SBP datasets. Together, these efforts aim to make sample data across the UMZH institutions findable and reusable, facilitating translational research and precision medicine initiatives.
Participating biobanks: USZ Tissue Biobank, USZ Bioresource (USZ), SCMB Biobank (Balgrist), multiple biobanks at PUK and Kinderspital Zürich, and clinical / research biobanks from the University of Zurich (Institutes of Medical Microbiology).
Partners: Swiss Biobanking Platform (SBP), The LOOP Zurich, the LOOP Biomedical Informatics Platform (BMIP), and the University Medicine Zurich (UMZH) network.
SeroAtlas
SeroAtlas is a Swiss-Belgian project that aims to reconstruct the immune landscape of infectious diseases between 2016 and 2025 using stored serum specimens from biobanks. By combining high-throughput multiplex serology with statistical modelling, the project will measure antibody responses to multiple pathogens from the same individuals and use these data to describe how immunity has evolved over time across age groups, and where possible by sex and region. It will identify population-level immunity gaps for pathogens with validated correlates of protection and assess whether antibody patterns can help predict subsequent seasonal or annual disease burden.
To achieve this, the project will harmonise and extend multiplex serology assays across laboratories in Belgium and Switzerland, process biobanked serum specimens, assemble linked and harmonised datasets, and develop statistical methods to generate representative immunity maps for both countries. Overall, SeroAtlas will deliver a retrospective serological atlas together with practical tools to support efficient serosurveillance and public health decision-making.
Applicants:
Isabelle Desombere (Sciensano, Belgium)
Niel Hens (University of Hasselt, Belgium)
Craig Fenwick (CHUV/University of Lausanne)
Julien Riou (Unisanté/University of Lausanne)
Partners:
Sabine Bavamian (SBP)
Joséphine Uldry (SBP)
Céline Pellaton (CHUV/University of Lausanne)
Mathieu Perreau (CHUV/University of Lausanne)
Andrew Azman (HUG/University of Geneva)
Start date is January 2027 (48 months).
Genome of Switzerland
The Genome of Switzerland (GoS) initiative aims to establish a national reference genomic dataset representing the genetic diversity of the Swiss population and providing a key resource for biomedical research and future genomic medicine in Switzerland. The project is designed to generate clinical-grade whole-genome sequencing (WGS) data from population-based samples collected through existing biobanks and cohorts, while ensuring compliance with national ethical and legal requirements as well as international standards for sample and data quality.
The pilot phase (2023–2025) focused on sequencing approximately 1,000 samples to generate the initial reference dataset and to establish the governance, ethical, technical, and analytical frameworks required for large-scale implementation. Building on this groundwork, a future scale-up phase aims to extend the dataset to up to 15,000 individuals and progressively integrate additional layers of molecular data (multi-omics), ultimately enabling Switzerland to contribute to international initiatives such as the Genome of Europe and strengthening the national ecosystem for genomic research and precision medicine.
The Genome of Switzerland (GoS) pilot brings together national infrastructures including the Health 2030 Genome Center, the Swiss Personalized Health Network (SPHN), the SIB Swiss Institute of Bioinformatics, Swiss Biobanking, and partners from the ETH Domain (ETH Zürich, EPFL and EMPA) through the Personalized Health and Related Technologies (PHRT) initiative, with links to the Swiss Multi-Omics Center. The contributing biobank is the Genomic Biobank from CHUV.
Collaboration with Swiss national research infrastructure
Swiss Clinical Trial Organisation (SCTO)
SCTO SBP survey
This survey aims to better understand current knowledge and practices related to sample management within the SCTO-CTU Network in order to strengthen its interaction with Swiss Biobanking Platform. In this survey, the term “sample management” is used as a generic term for biobanking.
The survey targets CTU staff who advise, coordinate, or support clinical trials or research projects involving biological samples. The results will help SBP and SCTO better understand CTU staff’s information and guidance needs regarding sample management and improve the support and collaboration Swiss Biobanking should provide to CTUs for clinical researchers.
Easy Guide to Clinical Studies (Easy-GCS)
The Easy-GCS is an online tool that provides researchers with easy accessible and relevant information – needed for study planning and conduct. Swiss Biobanking has worked in collaboration with SCTO to develop the biobanking module of the Easy Guide for Clinical studies.
The Easy-GCS is a comprehensive tool that provides all professionals involved in clinical studies answers and guidance on how to proceed with the set up and implementation of their study. In practice, the information related to the proper conduct of a clinical trial is accessible through a GRID divided into six study phases (from concept to completion) and eleven study subjects including the one focusing on Biobanking.
Swiss Cancer Institute: Memorandum of Understanding
The Swiss Cancer Institute and Swiss Biobanking recognize the growing importance of collaboration in advancing biomedical research and biobanking infrastructure. This Memorandum of Understanding (MoU) is intended to initiate a constructive dialogue and foster mutual understanding between the parties, laying out the groundwork for potential future cooperation.
This MoU establishes a preliminary understanding between Swiss Cancer Institute and Swiss Biobanking to explore potential collaborations and communication, including the exchange of information and ideas, with no binding obligations or commitments to proceed.
Parties agree to:
- Engage in discussions and sharing of non-confidential information related to their respective fields;
- Identify potential areas of mutual interest for collaboration;
- Consider possible projects, joint ventures, or research efforts on a non-exclusive basis;
- For identified opportunities create visibility in their communities on how collaboration could create mutual value.
SPHN: sounding board participation
The Swiss Personalized Health Network (SPHN) is operating a national infrastructure enabling multicentric research with health-related data. Launched in 2017 as an initiative by the Swiss government, SPHN is now on the way of becoming a sustainable part of the Swiss research infrastructure ecosystem according to the ERI Dispatch 2025-2028.
With this change in status came a change in governance structure. A new SPHN Sounding Board shall ensure the close alignment with the relevant stakeholders to stay connected to the network, remain informed, and advise the Steering Board and the SPHN Data Coordination Center on further developments of the network.
Tasks of Sounding Board members are:
- To receive and disseminate information from SPHN within their respective institution/organization
- To reflect on the strategy, guidelines, and implementation plans of SPHN
- To propose and endorse projects and working groups
- To support the acquisition of collaborations, mandates, and external projects
- To advise on fair distribution of costs and benefits within the network
- To improve conditions for the implementation of frameworks and partnerships.
The Sounding Board meets approximately twice a year with one representative for each institution listed below. For Swiss Biobanking, Christine Joye is the representative.
Annex 2: List of institutions to be represented in the SPHN Sounding Board
- Hospitals in SPHN
- Centre hospitalier universitaire vaudois (CHUV)
- Hopitaux universitaires Genève (HUG)
- Insel Gruppe AG
- Universitätsspital Basel (USB)
- Universitätsspital Zürich (USZ)
- Universitäts-Kinderspital Zürich
- Universitäts-Kinderspital beider Basel (UKBB)
- Kantonsspital Aarau (KSA)
- Kantonsspital Baden (KSB)
- Kantonsspital Luzern (LUKS)
- Health Ostschweiz (HOCH, St. Gallen)
- Ente Ospedaliero Cantonale (EOC, Bellinzona)
- The Hospitals of Switzerland (H+)
- Swissuniversities: Kammer Hochschulen
- Swissuniversities: Kammer Fachhochschulen
- Collège des doyens
- ETH Zurich
- EPF Lausanne
- Swiss Data Science Center (SDSC)
- H2030 Genome Center
- Swiss Academy of Medical Sciences (SAMS)
- SIB Swiss Institute of Bioinformatics
- Coordination Platform Clinical Research (CPCR)
- ORD Strategy Counsil
- Swiss National Science Foundation (SNSF)
- Innosuisse
- Swiss Clinical Trial Organization (SCTO)
- Swiss Biobanking Platform (SBP)
- Swiss Cancer Institute (formerly SAKK)
- Swiss School of Public Health (SSPH+)
- Patient partners
- Interpharma
- SwissMedTech
- The four SPHN & PHRT National Data Streams: IICU, LUCID, SPO-NDS, SwissPedHealth
- Federal Office of Public Health (FOPH)
- Federal Statistical Office (FSO)
- State Secretariat of Education, Research and Innovation (SERI)
BBMRI-ERIC's 10-Year Roadmap
The BBMRI‑ERIC 10‑Year Roadmap 2025–2035 sets a strategic vision for European biobanking, guiding collaboration, access to biological samples, and impactful research. Switzerland aligns these European objectives with national initiatives to strengthen biobanking across both levels. For each Strategic Objective outlined in the BBMRI Roadmap, dedicated Work Packages have been established to foster collaboration among all National Nodes, each comprising specific operational goals toachieve these objectives.
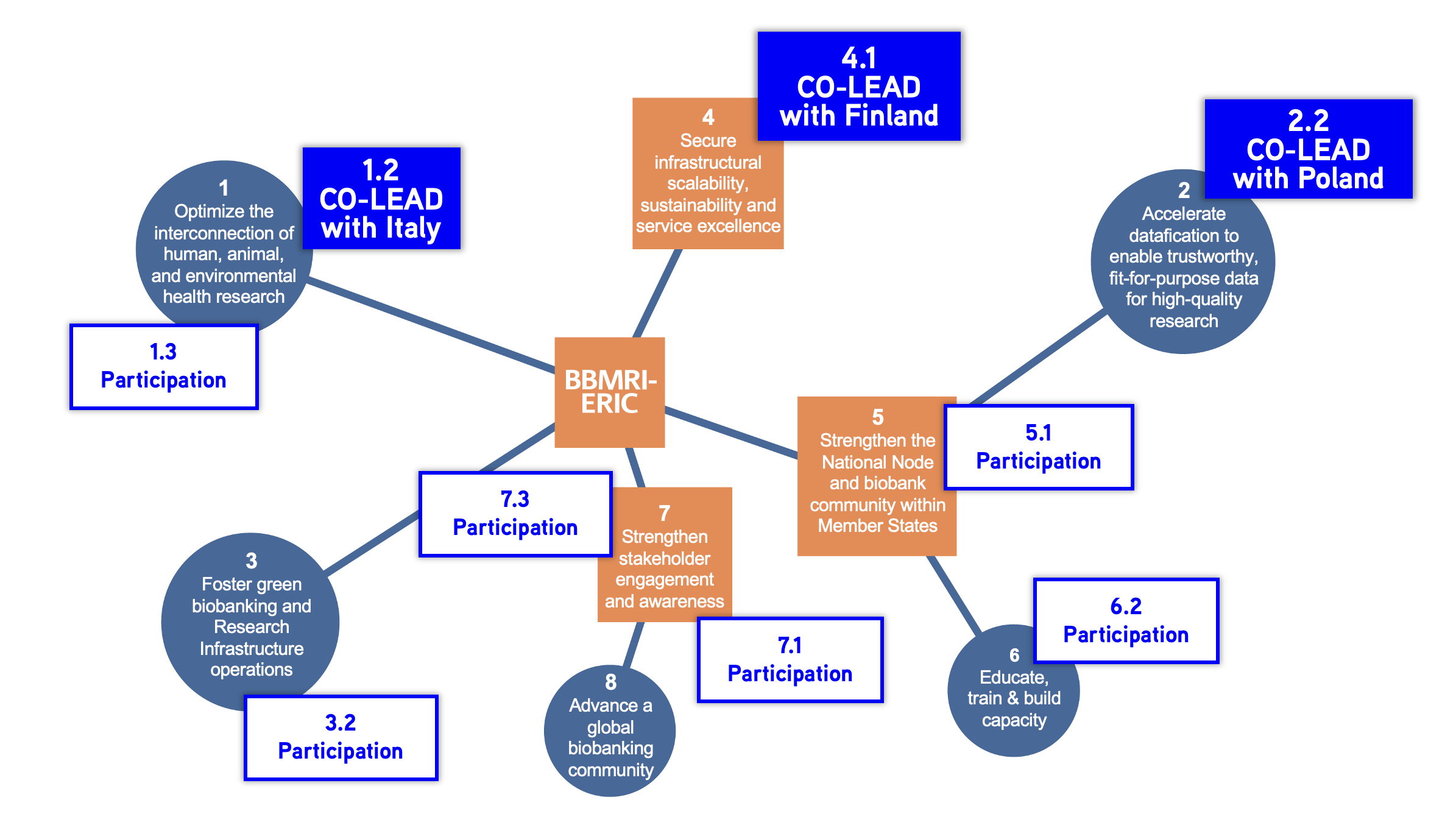
Objective SO1: Optimise the interconnection of human, animal, and environmental health research
BBMRI-ERIC will focus on optimizing the interconnection of human, animal and environmental health research by exploring opportunities based on the One Health paradigm.
SO1.2: Scale-up interactions and alliances with other research infrastructures and initiatives
Co-leads: CH (Joséphine Uldry & Christine Joye), IT (Marialuisa Lavitrano & Monica Forni)
Contributors NN: AT, BE, BG, CZ, ES, GR, IARC/WHO, LT, LV, NL, SI
In 2025, the SO1.2 group held several co-lead and participant meetings to coordinate cross-country efforts and refine its strategic approach to exploring potential interactions and alliances across Europe in the One Health domain. A flexible survey was launched in collaboration with SO1.3 to map non-human networks and research infrastructures, with participation from six countries. The mapping identified key domains across national and EU landscapes, including data and bioinformatics, infectious disease and microbial resources, environmental and ecosystem research, veterinary and animal health, and social sciences. It confirmed the existence of a broad but fragmented One Health ecosystem and revealed substantial expertise within the BBMRI network. Survey responses indicated that reported links between non-human initiatives and biobanking are primarily project-based, with limited structural integration, particularly in environmental and biodiversity domains, providing important insights for future strategic development.
In parallel, the group initiated work on the ELSI of One Health, including the preparation of a dedicated ELSI dialogue on the role of biobanks in realizing the One Health approach, involving renowned experts from the field.
Objective SO2: Accelerate datafication to enable trustworthy, fit-for-purpose data for high-quality research
Accelerating datafication by applying FAIR-Health principles will enable reproducible advancements in science. As FAIR, is relates to Findable, Accessible, Interoperable, Reusable, quality-defined, with incentives for data sharing and reuse, including ELSI aspects (Ethical, Legal and Societal Issues).
SO 2.2 Enable reproducible research through good data traceability and quality management
Co-leads: CH (Joséphine Uldry & Flavien Delhaes), PL (Agnieszka Matera-Witkiewicz & Błażej Marciniak)
Contributors NN: AT, DE, ES, GR, HU, IT, NL.
In 2025, the SO 2.2 group held multiple co-lead and participant meetings to exchange expertise on provenance and traceability, coordinate cross-country efforts, and refine the strategic approach for implementing a provenance model across the BBMRI-ERIC network.
A comprehensive status analysis was conducted to assess readiness levels among National Nodes and affiliated biobanks regarding sample and data traceability, provenance implementation, and quality management (QM) maturity. Individual readiness scores were calculated for each biobank, enabling the development of a landscape overview reflecting traceability capacity and QM maturity across participating countries.
A key activity was the identification of biobanks suitable for piloting a provenance infrastructure under the EvolveBBMRI framework. Ten advanced biobanks from six countries were evaluated with strong digital capacity and quality management alignment.
Major achievements include the identification of key bottlenecks, such as limited linkage across data type and limited automation and IT system integration, and the definition of concrete priorities for 2026. The structured, maturity-based approach to provenance implementation and the selection of pilot biobanks represent significant progress toward a harmonized and digital biobanking across Europe.
Objective SO4: Strategic Objective: Secure infrastructural scalability, sustainability and service excellence
To further enhance the infrastructural scalability, sustainability and service excellence, we seek to engage with universities, industry and private foundations to explore collaboration models such as support for specific visionary projects, in-kind contribution, industry collaboration, public-private partnership as well as open innovation models. In parallel, the ambition is also to nurture close and high-value partnerships with associated and third countries.
SO 4.1 Explore and develop new services models
Co-leads: FI (Tom Southerington), CH (Sabine Bavamian & Khalil Roy & Lou Ferraton)
Contributors NN: AT, CZ, DE, ES, GR, SE.
In 2025, activities under SO4.1 focused on exploring and developing new service models to strengthen collaborations and improve service provision across the BBMRI-ERIC network. Two main objectives guided the work.
First, efforts were dedicated to developing a legal framework enabling collaborations across the BBMRI community (biobanks, National Nodes and HQ) and with external partners, particularly industry. A concept for a collaboration model was developed in which BBMRI-ERIC would act as a single-entry partner for industry, channeling requests for samples and data to relevant partners within the BBMRI network. The objective of this “one-stop shop” model is to enable faster responses to industry needs while establishing contractual and governance frameworks in advance to facilitate efficient service delivery. A pilot with an industrial partner is currently explored and a draft collaboration agreement was initiated. Ongoing discussions still need to carefully address contractual obligations, liabilities and performance expectations between BBMRI, the service provider and the industrial partner(s).
Second, SO4.1 worked on defining the concept and requirements for a universal, user-friendly platform providing a central entry point to explore and access biological resources, services and other relevant information (e.g. trainings) across the BBMRI community. User needs were identified and the platform concept is currently under development.
European projects
Evolve BBMRI-ERIC Federated Platform project
The EvolveBBMRI project brings together a consortium composed of 11 partners: BBMRI-ERIC as the coordinator and 10 National Nodes. The overarching goal of the project is to (together with the Member States) further develop BBMRI-ERIC for the benefit of its scientific communities and users, increase its capacity to serve EU policy priorities and further deepen its collaboration with industry.
Work Package 1 “Acceleration of datafication of biobanks and biomolecular resources to enable reproducible advanced medical research”, is in line with the Strategic Objective 2 “Accelerate datafication to enable trustworthy, fit-for-purpose data for high-quality research” of BBMRI-ERIC’s 10-Year Roadmap for 2025-2035. Within WP1, a call for interest to join the BBMRI-ERIC Federated Platform (e.g. Federated search and provenance mechanism) has been launched to enhance the availability and quality of biomolecular resources for the research community.
In Switzerland, Swiss Biobanking applied and has been selected to the Evolve call for the Federated Platform to be the single entry point through the SBP NExT and ease onboarding of CH Biobanks part of SBP network.
As single biobanks often don’t have the knowledge and manpower to develop such projects or don’t have access to IT specialists, SBP applies to this call to provide biobanks with an easier way to get access to the Federated Platform with minor investments for them. By integrating your biobanks with the BBMRI-ERIC Federated Platform, it will enhance international visibility, expand your reach to a wider audience, and open doors to global collaborative projects.
Project duration: 6 months (March to August 2026)
Beneficiaries in CH: Swiss Biobanking
Contact persons: Sabine Bavamian, Raphaël Colsenet and Flavien Delhaes
CaBRIOH Capacity Building in Research Infrastructures towards One Health (HORIZON-INFRA-2025-01)
Pandemics, antibiotic resistance, climate change, and deforestation have clearly exposed a bitter truth: the health of humans, animals, plants, and ecosystems is inextricably linked. The One Health paradigm calls for an integrated response, but its success depends on a workforce of technical professionals on all maturity levels that can connect science, infrastructure, and policy.
Yet training for this essential workforce remains fragmented, undervalued, and rarely portable across borders. Without a coordinated system for competence and skills development and recognition, Europe risks losing ground in scientific excellence, research innovation, and global competitiveness.
The CaBRIOH project unites leading actors across the ESFRI Health & Food domain. At its core is BBMRI-ERIC with BBMRI.it, BBMRI.at, BBMRI.ch (Swiss Biobanking), BBMRI.de and BBMRI.es, focusing on human and animal health. This foundation is complemented by EIRENE RI, with environmental exposure and human exposome research. Together, these partners provide comprehensive coverage of the One Health agenda.
CaBRIOH’s approach is participatory (co-designed with RI staff and stakeholders), interoperable (aligned with EQF, ESCO, EOSC, EHDS2), and scalable (embedded in BBMRI-ERIC and transferrable to other ESFRI initiatives). The project has two hearts that beat as one: the extensive provision of high-quality training modules—including mobility schemes that link RIs across countries and domains—and the NextGen training platform, a one stop hub that consolidates all offers, personalizes learning pathways, and ensures skills are recognised through micro-credentials, Europass and EU Digital Identity Wallets. Together, they guarantee that competencies are not only acquired but also visible, transferable, and impactful across Europe and beyond.
In Switzerland, the first training program, called the CAS in Biobanking (Certificate of Advanced Studies) launched in 2025, is part of the initial training landscape in Europe. The CaBRIOH project will enable to upskill any professional acting in biobanking by identifying areas of expertise missing and developing new education contents.
Project duration: 30 months (June 2026 to December 2028)
Beneficiaries in CH: Swiss Biobanking
Contact: Sabine Bavamian, Valeria Di Cola and Adam Strobeyko
Swiss Cooperation programs
Czech-Swiss Biobank Infrastructure Initiative for FAIR Data and Qualtiy Certification (CS-BIQ)
Call 8K2502 for proposals in bilateral collaboration between Czech and Swiss research infrastructures within the Czech-Swiss Research Infrastructure Initiative with project duration from 2025 to 2028.
The CS-BIQ project connects the national nodes of BBMRI-ERIC in Czechia (BBMRI.cz) and Switzerland (BBMRI.ch) with the aim of strengthening the quality and interoperability of biobanking services in both countries. It focuses on two key areas: i) harmonisation of biospecimen quality in biobanking processes, and ii) institutional readiness for FAIRification of data associated with biospecimens.
In the area of quality, a swiss evaluation system will be introduced to support Czech biobanks to obtain the internationally recognised BBMRI-ERIC Quality Label or ISO 20387 accreditation. The Swiss partner will provide methodological guidance and training, while the Czech node will gradually take over the initiative of evaluation in the project lasting period (WP 1).
The project will support data integration and cataloguing on site resulting in metadata findable in the national catalogues of BBMRI.cz (data.bbmri.cz) or BBMRI.ch (SBP NExT). The goal is to automatise metadata management and preparation to set FAIRification processes in the biobanks for biospecimens and data (WP 2).
The project will develop an ethical and legal framework for sharing biospecimens and data between Czechia and Switzerland to simplify international collaboration in compliance with GDPR and Swiss regulations. This framework will be helpful for future cohort development when a demand will come (WP 4).
The project will strengthen the capacities of both national nodes, establish a reusable model of collaboration, and increase the visibility, and efficiency of biobanks in both countries.
Project duration : 36 months (March 2026 to February 2029)
Beneficiaries in CH : Swiss Biobanking
Contact persons : Sabine Bavamian, Raphaël Colsenet and Flavien Delhaes
Swiss-Hungarian Cooperation Programme II, “Research and Innovation” thematic area, SM02-RES-PC3-AB and SM02-RES-PC3-C, entitled ”Support for Cooperation between Research Infrastructures “ for the purpose of selecting beneficiaries. SM02-RES “Research programme Hungary” PC3
Developing Swiss–Hungarian Federated Data Sharing to Advance the BBMRI-ERIC
OneHealth Biobanking Concept
Swiss and Hungarian partners collaborate to implement federated data sharing, combining complementary strengths and diverse genomic resources through demonstration studies in human and plant genomics, aligned with the One Health concept
This federated, privacy-preserving technology enables the extraction and analysis of valuable information from individual records without central aggregation, thereby ensuring security and confidentiality of sensitive data. In human genomics, the project focuses on hereditary cancer syndromes, pharmacogenetic variation, and actionable secondary findings—genetic variants not linked to the primary condition but detectable through comprehensive testing and relevant for prevention or treatment. Analysis of cancer-predisposing genes estimates variant frequency and risk, guiding precision screening and personalized surveillance. Population level assessment of pharmacogenetic variants supports safer and more effective medication use. Evaluation of medically actionable variants according to ACMG guidelines promotes responsible return-of-results policies and clinical translation.
Pilot studies in plant genomics show how federated infrastructures can integrate human, plant, and environmental datasets within one platform. Case studies with high scientific value and translational relevance address actionable genetic risks and deliver benefits for patients, healthcare systems, and ecosystem health.
Overall, the project validates cross-border federated data sharing and demonstrates the potential of interoperable genomic resources to advance precision medicine and One Health research, creating a secure framework that strengthens healthcare and supports sustainable health for humans, plants, and the environment.
Modern science is moving toward data-driven, personalized and sustainable health, spanning humans, plants and the environment. This enriches biobank data with multidimensional context for precise prevention and treatment. Achieving this requires secure, interoperable integration of genomics, exposomal and clinical data.
Biobanks are central, but their potential depends on: 1.) linking and harmonizing data across institutions; 2.) GDPR-compliant sharing; 3.) enabling AI research without centralizing sensitive data. Federated learning meets these needs by enabling collaboration without data transfer, ensuring privacy, compliance, and alignment with EU initiatives like the EHDS.
Harmonised genomic data allow estimation of population-specific inherited risk variants, focusing on actionable and preventable conditions, while One Health integration links human genomics with plant data such as the grapevine genome. This bilateral project leverages complementary expertise and diverse resources, enabling countries to learn from each other’s datasets and experiences.
It delivers secure computation and harmonisation standards, addresses priorities including oncology and pharmacogenomics, and facilitates legal alignment, trust, and One Health integration by linking human with plant genomic data. Together, these activities validate cross-border federated data sharing and highlight the potential of interoperable genomic resources to advance precision medicine and sustainable health across species and systems.
Project duration : 36 months (March 2026 to February 2029)
Beneficiaries in CH : Swiss Biobanking
Partners in CH : Swiss Data Science Center and Genome Center
Contact persons : Sabine Bavamian, Joséphine Uldry and Adam Strobeyko

