Labels
Our Labels recognize Swiss biobanks that comply with applicable legal and ethical requirements and apply best practices
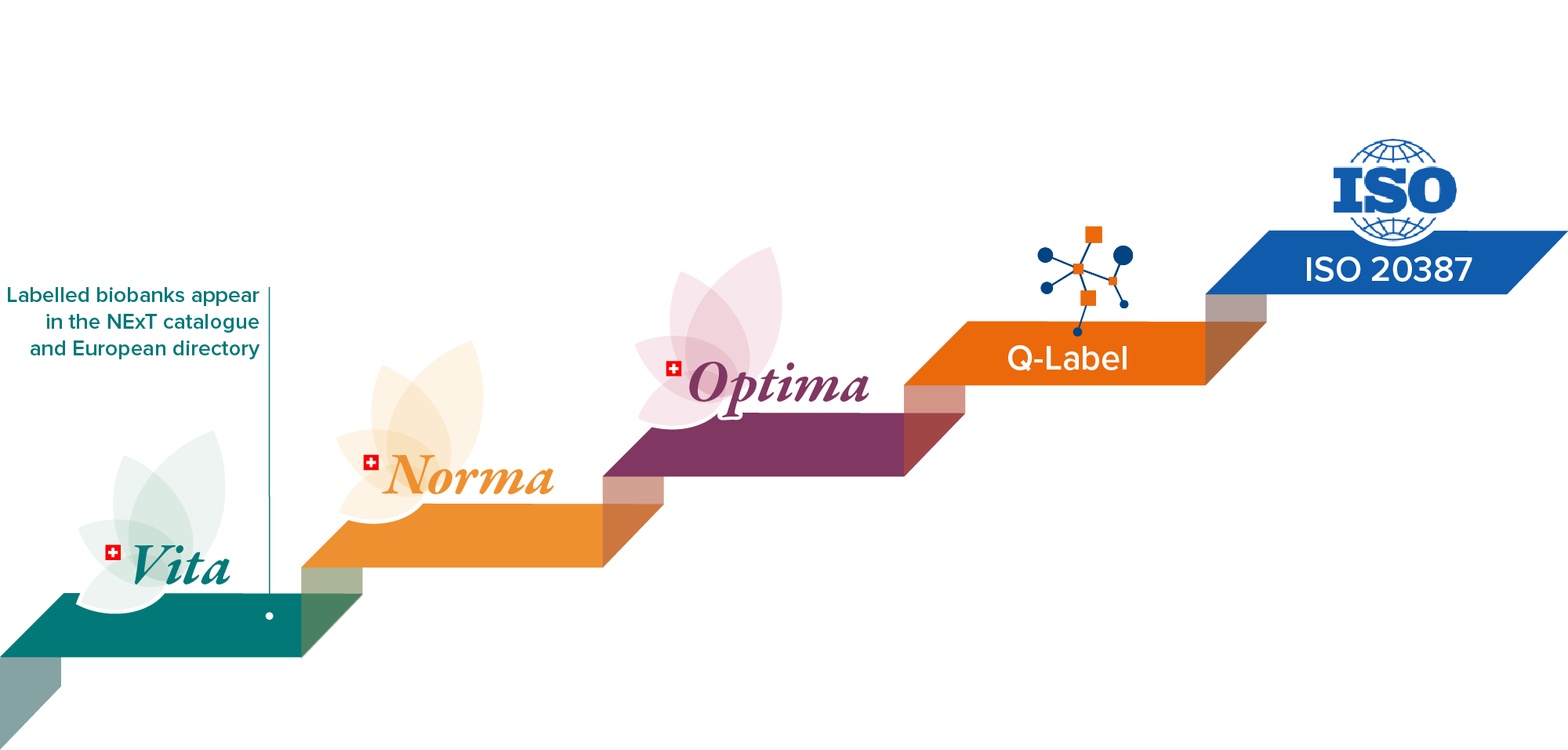
Recognizing quality and strengthening trust in Swiss biobanking. Our Labelling Program provides a stepwise approach that helps biobanks achieve and demonstrate their compliance with standards in quality, and set up appropriate governance. The three levels — VITA, NORMA, and OPTIMA — reflect increasing degrees of operational maturity and support different stages of development.

The Labels help biobanks establish their practices, align with recognized standards such as ISO 20387, and document key steps of the pre-analytical phase. Each level includes a structured evaluation of the biobanking activities and provides the necessary support. Labelled biobanks appear in the NExT catalogue and European directory, increasing their visibility to researchers to foster collaborations.
Key benefits
- Legal and ethical support to set up a compliant biobank
- Demonstrate your conformity with the applicable framework
- Benefit from documentation (incl. templates) and support addressing legal and ethical requirements
- Good practices development to improve quality
- Follow structured evaluation
- Use template documentation (e.g. SOPs, forms, records) to develop a proper practice
- Preparation for ISO20387 standards
- Foster harmonized practices supporting ISO readiness
- Progress step-by-step through the Label Program
- Increased recognition and visibility
- Become visible nationally (Swiss Network) and in Europe (BBMRI-ERIC Network)
- Connect with the biobanking Community
Who is it for?
The Labels are designed for all human and non-human biobanks collecting, storing, or managing biological samples for research, whether institutional, hospital-based, or independent (public or private). They are suitable for biobanks at any stage of development, from newly established entities to mature infrastructures seeking to align with national and international standards.
The process is progressive and adaptable, making it accessible across diverse operational contexts.
Director of the Applied Microbiology Research Group University of Zurich
and Vice-President of Swiss Biobanking Platform
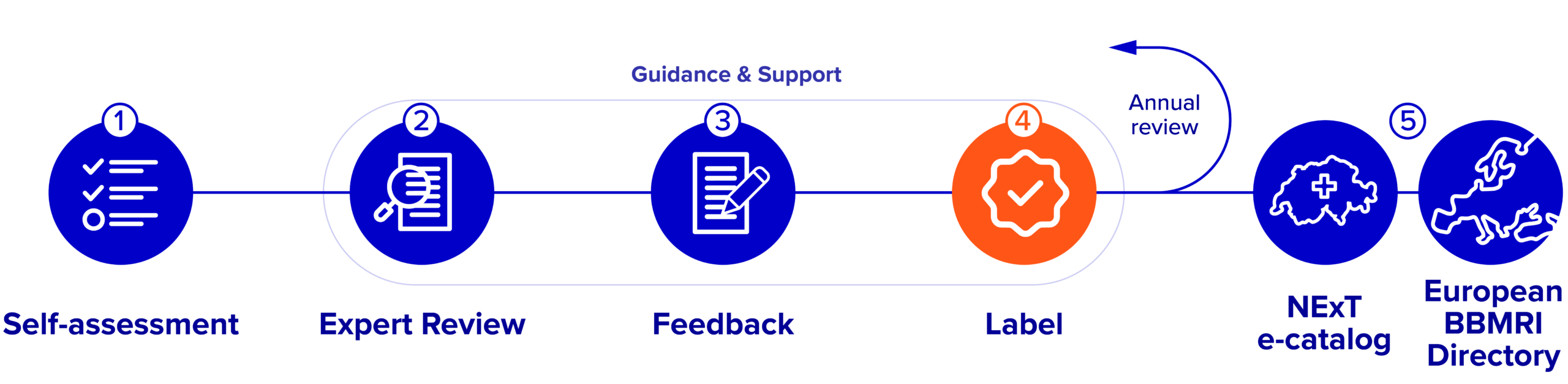
How to obtain a Label
-
Registration & self-assessment
Biobanks register on our online evaluation platform and complete a detailed questionnaire covering governance, processes, and Quality Management System (QMS). -
Expert review
Document review for all labels followed by an online meeting for the VITA Label or an on-site visit for NORMA and OPTIMA Labels. -
Feedback
A report to summarize our observations, exchanges and recommendations based on identified gaps, along with targeted support material (e.g., SOPs templates). -
Label award & annual follow-up
Successful biobanks receive the Label; yearly reviews ensure continued compliance and up-to-date information. - Directory listing
Labelled biobanks are eligible to appear in the Swiss (NExT) and European (BBMRI-ERIC Directory) directories, increasing their visibility within the research community.

VITA Label
Compliance with the legal and ethical framework
The VITA Label recognizes biobanks that meet requirements in governance aspects. It is awarded through a remote evaluation based on a document review (e.g. biobank regulation, consent documents, etc).
Biobanks with the VITA Label are listed in the NExT national sample catalogue, increasing their visibility to researchers.
Governance
Regulation, agreements, consent
Compliance
Responsibilities, ethics, confidentiality, safety
Communication
Results, external communication, complaints
- MTA 2.0 (Template)
- MTA 3.0 (Template)
- MTA Veterinary (Template)
- Data Quality SOP (SOP)
- Responsibilities SOP (SOP)
- Safety and Complaint SOP (SOP)
- Obtaining consent SOP (SOP)
- Participant Identification log (Template)
- Ethical legal and professional compliance list (Guideline)
- Biobank regulation (Template)
→ View all documents in the document library

NORMA Label
Standardization of the operational processes
The NORMA Label evaluates the quality of the operational processes and increases the harmonization of practices. Unlike the VITA Label, SBP examines the practices and processes of your biobank during an on-site visit.
Resources & infrastructure
IT systems and security, traceability, equipment, maintenance, critical consumables
Personnel & competencies
Roles and responsibilities, job descriptions, training
Process management
Collection, transport, reception, processing, storage and distribution processes
Methods & quality control
Validated methods, quality control procedures and results
- Datasets Bacteria (Dataset)
- Datasets Veterinary (Dataset)
- Datasets Human (Dataset)
- Quality control strategy implementation SOP (SOP)
- Validation of methods SOP (SOP)
- Biological Material Management SOP (SOP)
- Personnel management SOP (SOP)
- Equipment Management SOP (SOP)
→ View all documents in the document library

OPTIMA Label
Optimization of the Quality Management System
The OPTIMA Label allows your biobank to establish a QMS with proper documentation management and implementation of Quality Indicators to monitor and optimize your operational processes. This highest level supports long-term sustainability and continuous improvement.
Quality governance
Quality policy, service level agreements, internal communication
Documentation & information management
Document management system, document control and lists
Continuous improvement
Management reviews, satisfaction surveys, internal audits, nonconformities management
Risk & sustainability
Biosafety and risk management, business model and cost recovery
- Service Level Agreement Template (Template)
- Risk Management SOP (SOP)
- Internal audit SOP (SOP)
- Documentation management SOP (SOP)
- Improvement Management SOP (SOP)
- Non-conformity management SOP (SOP)
- Quality Manual (Template)
→ View all documents in the document library
— Dr Irina Banzola
Urology Clinic, University of Zurich
FAQ
SBP Labels are about:
- Ensuring that your biobank fulfills legal and ethical requirements
- Receiving support and training in the implementation of Good Biobanking Practices, so that samples and data are fit for research purposes
- Joining a community: SBP Labels provide access to the SBP network, enabling members to collaborate and effectively advocate for shared interests
- Increasing visibility of your samples for researchers by being integrated in NExT, our Swiss catalogue, and the largest European one (BBMRI-ERIC directory)
- Building trust with all stakeholders, including researchers and participants
Yes! SBP Labels are designed for all types of biobanks, regardless of size or sample type.
This includes human samples (e.g., liquid, tissue), non-human samples (e.g., microbiome, animal, plant), as well as biobanks ranging from national population cohorts to rare disease biobanks to natural history museum collections or pathogen biobanks.
SBP Labels support and train biobanks to improve quality by providing guidance, training, and evaluations aligned with ethical, legal, and good biobanking practices.
ISO 20387, on the other hand, is a formal accreditation issued by an external body to confirm full compliance with international standards. SBP Labels support biobanks towards this path but are not formal ISO accreditations themselves.
SBP VITA Label: The criteria follow Swiss laws (such as the Human Research Act and Human Research Ordinance) and international guidelines like the Declaration of Taipei. SBP also includes its own recommendations, for example on the minimal documentation of pre-analytics or on data management.
SBP NORMA and OPTIMA Labels: The criteria are based on widely recognized international standards and guidelines for biobanks, including OECD, IARC, and ISBER recommendations, as well as ISO standards for biobanking and quality management (mainly the ISO 20387). SBP also includes its own recommendations, for example on traceability.
A QMS helps biobanks do the right things in the right way, ensuring that the needs of researchers and participants are met. It standardizes processes, making work more efficient, consistent, and reliable, while reducing errors. Standardization also makes it easier to train staff, share data, collaborate with other biobanks, and increase quality of your operations. Quality checks and good documentation help biobanks manage their activities effectively and continuously improve their practices.
We need two weeks to review the documents after receiving them. Then we need two weeks after the online or on-site compliance review to write the report and deliver the Label if biobank is deemed eligible / time depends as well on how much time the biobank needs to prepare all documents.
The Label status is currently visible on the SQAN (online questionnaire) and on NExT. Biobanks are encouraged to feature it on their own websites and other supports using our communication kit and receive a paper certificate for presentation. The labeling is also promoted through our social media channels.
Contact expert
Josephine Uldry
Quality Officer — Human and non-human
josephine.uldry@swissbiobanking.ch
Lou Ferraton-Walder
Project & Operations Officer — Human
lou.ferraton@swissbiobanking.ch

